|
6/22/2023 0 Comments Hexane density
For a given molar mass, the boiling points of alkanes are relatively low because these nonpolar molecules have only weak dispersion forces to hold them together in the liquid state. Larger molecules have greater surface areas and consequently interact more strongly more energy is therefore required to separate them. This general rule holds true for the straight-chain homologs of all organic compound families. Neither ILO nor WHO nor the European Commission shall be responsible for the interpretation and use of the information contained in this material.\) are the boiling points of the straight-chain alkanes increase with increasing molar mass. The published material is being distributed without warranty of any kind, either expressed or implied. The substance is toxic to aquatic organisms.ĭepending on the degree of exposure, periodic medical examination is suggested. MAK: 180 mg/m 3, 50 ppm peak limitation category: II(8) pregnancy risk group: C. Animal tests show that this substance possibly causes toxic effects upon human reproduction. The substance may have effects on the central nervous system and peripheral nervous system. Repeated or prolonged contact with skin may cause dermatitis. Exposure at high levels could cause lowering of consciousness.Ī harmful contamination of the air can be reached rather quickly on evaporation of this substance at 20☌.Įffects of long-term or repeated exposure If this liquid is swallowed, aspiration into the lungs may result in chemical pneumonitis. at 254nm: Color: Colorless: Assay Percent Range. Specifications Melting Point-95☌: Absorbance: 1.00 AU max. The substance can be absorbed into the body by inhalation of its vapour and by ingestion. hexane: SMILES: CCCCCC: Description HPLC mobile phase for analytical and preparative separations. Octanol/water partition coefficient as log Pow: 3.9 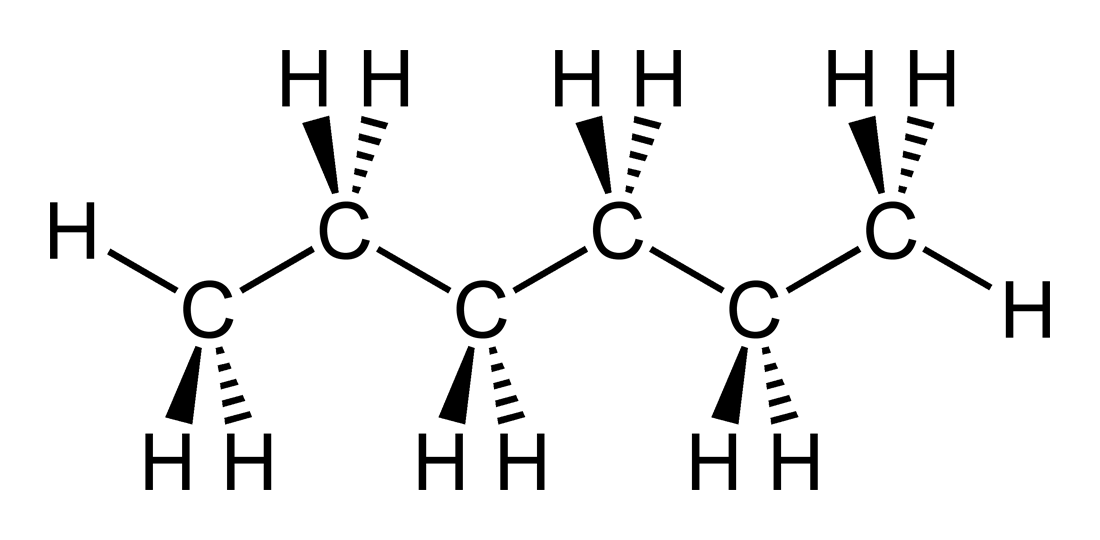
Relative density of the vapour/air-mixture at 20☌ (air = 1): 1.3 Attacks some plastics, rubber and coatings. This generates fire and explosion hazard. The vapour is heavier than air and may travel along the ground distant ignition possible. VOLATILE COLOURLESS LIQUID WITH CHARACTERISTIC ODOUR. With the financial assistance of the European Commission. Prepared by an international group of experts on behalf of ILO and WHO, Critical density Data from NIST Standard Reference Database 69: NIST Chemistry WebBook The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific judgment. 
Then store and dispose of according to local regulations.įireproof. Absorb remaining liquid in sand or inert absorbent. Collect leaking and spilled liquid in sealable containers as far as possible. Do NOT let this chemical enter the environment. The demixing pressures, densities and viscosities of solutions of nearly monodisperse polystyrene samples (Mw/Mn < 1.1) in n-hexane have been determined. Ĭonsult an expert! Personal protection: filter respirator for organic gases and vapours adapted to the airborne concentration of the substance. Wear safety goggles, face shield or eye protection in combination with breathing protection.įirst rinse with plenty of water for several minutes (remove contact lenses if easily possible), then refer for medical attention. 
Rinse and then wash skin with water and soap. Use ventilation, local exhaust or breathing protection.įresh air, rest. In case of fire: keep drums, etc., cool by spraying with water.ĭizziness. Do NOT use compressed air for filling, discharging, or handling. Closed system, ventilation, explosion-proof electrical equipment and lighting. NO open flames, NO sparks and NO smoking.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
